|
2/10/2024 0 Comments White precipitateThe cyanuric acid has the physical properties of being slightly soluble in cold water and easily soluble in hot water. After long time, a large amount of crystals are generated in the pipeline and urea tank of the SCR system.ĪdBlue ® for diesel vehicles will heat at high temperature during use, and the triuret will be condensed into cyanuric acid, which will clog the AdBlue ® nozzle. Tell students that a chemical reaction took place and that a solid was formed. The white precipitate formed in this test is due to the presence of water. These white precipitates can cause the malfunction of AdBlue ® detector.Īt room temperature, the dehydration and crystallization of triuret causes the AdBlue ® sensor of the vehicle to malfunction, the AdBlue ® level detection is inaccurate. White solid particles will form and deposit as a precipitate. white precipitate test 5 Aqueous sodium hydroxide was added dropwise and then in excess to the fourth portion of solution E in the boiling tube. The density of triurea is relatively large, and it will accumulate and precipitate to the bottom.ġ. white precipitate test 4 Excess aqueous ammonia was added to the third portion of solution E. Through the professional detection of AdBlue ® with precipitate after melting, the test result shows that the main component of the white precipitate is triuret, the solubility of triuret under low temperature conditions is very low, low temperature leads to precipitation. The crystals are more dense than the surrounding solution and rain down, creating a trail of white silver chloride behind as a trace of the precipitate's path. Under the right conditions, small crystals of silver chloride form within the cloud. The AdBlue ® in urea tank melt after freezing, white sediment will appear at the bottom, so the low temperature is one of the conditions for the formation of sediment. The bulk of the precipitate appears in the form of a suspended cloud of white solid. White precipitate which dissolves to form a colourless solution when excess NaOH solution is added and when excess NH 3 solution is added.The white precipitate in AdBlue ® usually appear in winter. Chloride With silver nitrate TS, solutions of chlorides yield a white, curdy precipitate that is insoluble in nitric acid but is soluble in a slight excess of 6 N ammonium hydroxide.When testing amine (including alkaloidal) hydrochlorides that do not respond to the above test, add one drop of diluted nitric acid and 0. White precipitate forms which dissolves to form a colourless solution when excess NaOH solution is added but the white precipitate remains when excess NH 3 solution is added. White precipitate which remains when excess NaOH solution or NH 3 solution added. Green precipitate which remains when excess NaOH solution or excess NH 3 solution added.īrown precipitate which remains when excess NaOH solution or excess NH 3 solution added. Trending now This is a popular solution Step by step Solved in 2 steps with 2 images. Make into a solution and add an excess of NaOH solution or NH 3 solution. The solid that separates is called a precipitate. When dilute HCl is added to a solution, a white precipitate forms. Be (OH) 2, Mg (OH) 2 are insoluble in water. 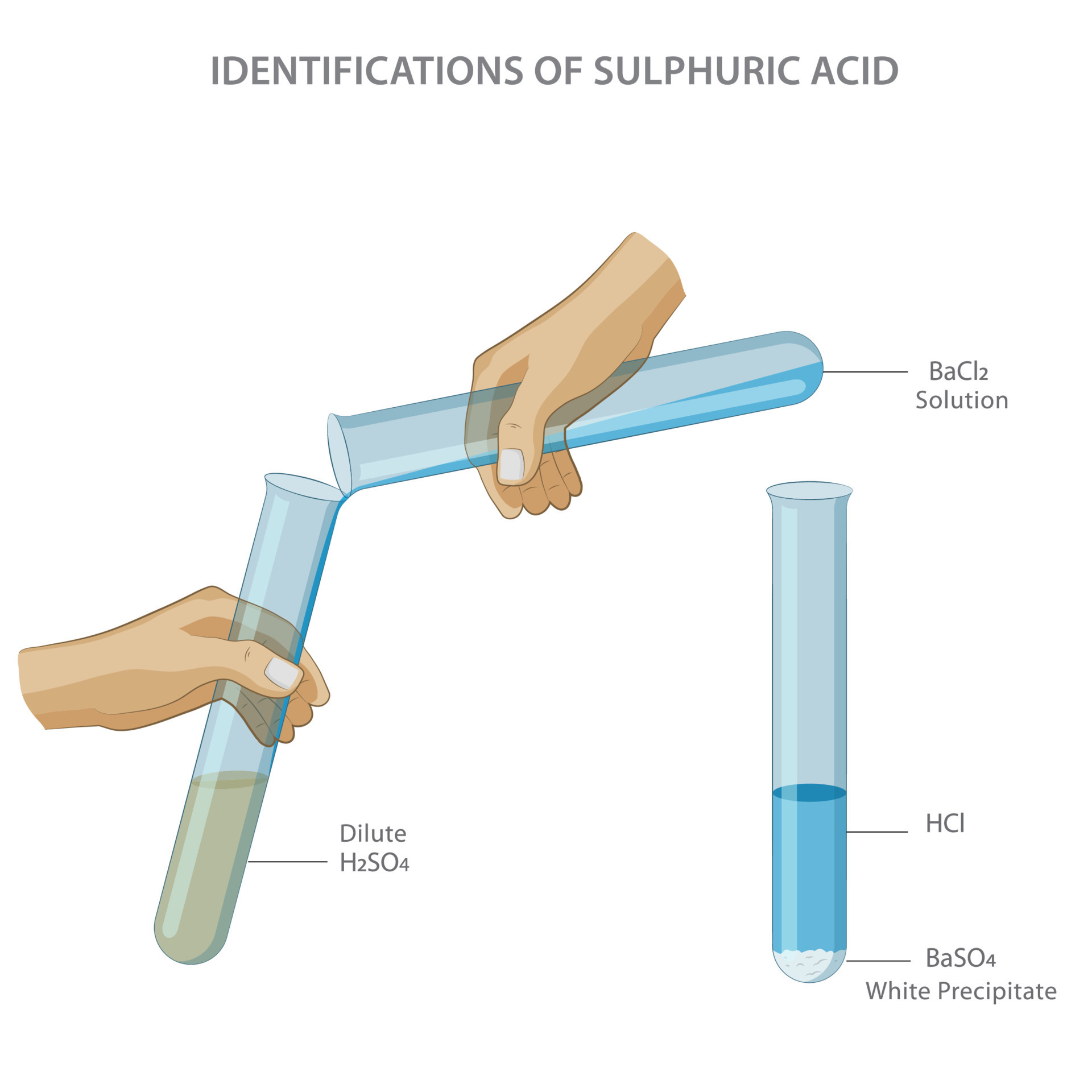
SrSO 4, BaSO 4 are white colour precipitates.  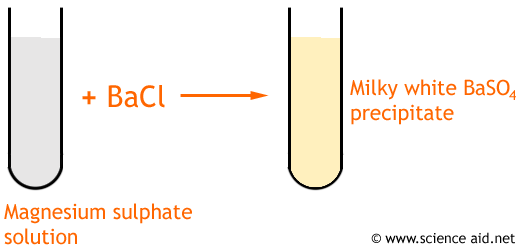
You can quickly check on the effect by preparing a 0,4-0,5 mM solution of. CaCO 3, MgCO 3, SrCO 3 are precipitates and they are white. The white precipitate is indeed caused by the imidazole itself and leaked Ni2+-ions from the metal-affinity-column. All carbonates (CO 32-) of alkali earth metal ions are precipitates. Make into a solution and add an excess of NH 3 solutionīlue precipitate which remains when excess NaOH solution added.īlue precipitate which dissolves to form a deep blue solution when excess NH 3 solution added. Alkaline earth metals forms both precipitates and solutions. Dissolve in water, add a few drops of NaOH solution, or NH 3 solution, and continue to add until it is in excess
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
